|
1/23/2024 0 Comments Ph3 molecular geometry bond angle 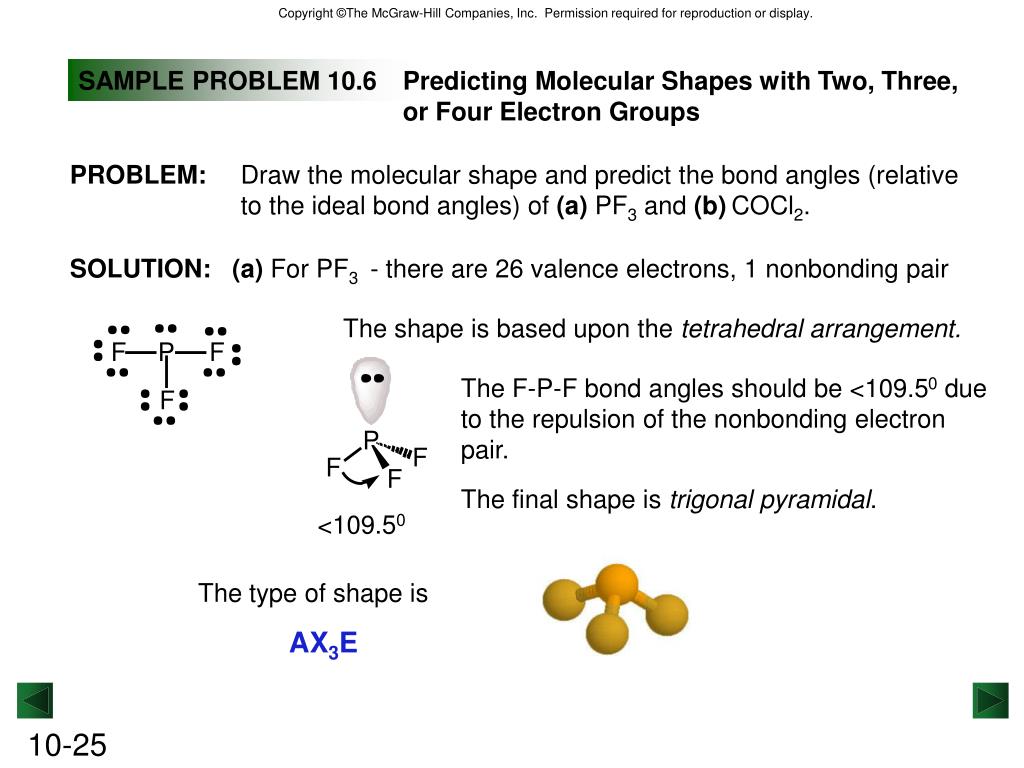
The geometries of molecules can be calculated by the number of lone pairs and bond pairs in the molecule. This provides the shapes and geometries to the various molecules. Due to the repulsion of electrons in the molecule, atoms move far apart and occupy a position in the space that provides minimum repulsion for the molecule. This results in a greater effect of lone pair repulsions.ħ. 
Bond pair electrons are those shared between two atoms where the electron density is distributed between two atoms, and the effect decreases. Lone pair repulsions are stronger than the other repulsive forces because lone pairs are present on the central atom and occupy greater space. The order of repulsive forces is bond pair- bond pair repulsions Repulsions can be of different types, like bond pair-bond pair repulsions, lone pair-bond pair repulsions and lone pair-lone pair repulsions.Ħ. Since all the electrons are the same when they are closely placed, they undergo repulsion. Bonded pairs of electrons are those which are shared between two atoms in a molecule, whereas lone pairs of electrons are present only on a single atom that is the central atom.ĥ. Valence electrons can be mainly of two types – bonded electrons and lone pair electrons.Ĥ. In an element, the electrons present in the outermost shell are only responsible for bond formation. The element can combine with other elements to attain stability by sharing electrons.Ģ. This theory is mainly proposed to explain the geometries, shapes and bond angles of the molecules based on valence electrons in the central atom. Valence Shell Electron Pair Repulsion Theory (VSEPR): To explain molecular geometry and bond angles, the most important theory we need to discuss is the VSEPR theory. According to different theories, the arrangement of bonds around the central atom can be known through which bond angles are explained. Molecules forming a covalent bond with different atoms usually are arranged in various forms. Molecules forming a covalent bond with the same atoms with more than two atoms are polyatomic molecules for example, O3, P4, S8. Molecules forming a covalent bond with the same atoms, like homoatomic molecules, are usually arranged linearly for example, H2, I2, Br2, Cl2, F2 etc. The covalent bond is usually formed by sharing electrons, without ions and are directional. The ionic bond is formed by the transfer of electrons, forms ions and is crystalline and non-directional. Ionic and covalent molecules are very different from one another. Molecules have atoms bonded around them that occupy space and can have different bonds like covalent and ionic. Compounds have a fixed composition irrespective of the source from which they are prepared, and this follows the law of constant proportion. Molecules that are made of two atoms are binary compounds and those made of three atoms are tertiary compounds. Molecules can be diatomic, triatomic or polyatomic and can be made of the same type of atoms or different types of atoms. Inert gases or noble gases are stable, but the rest of the elements are not stable. Elements that are naturally unstable combine with other elements to gain stability. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
